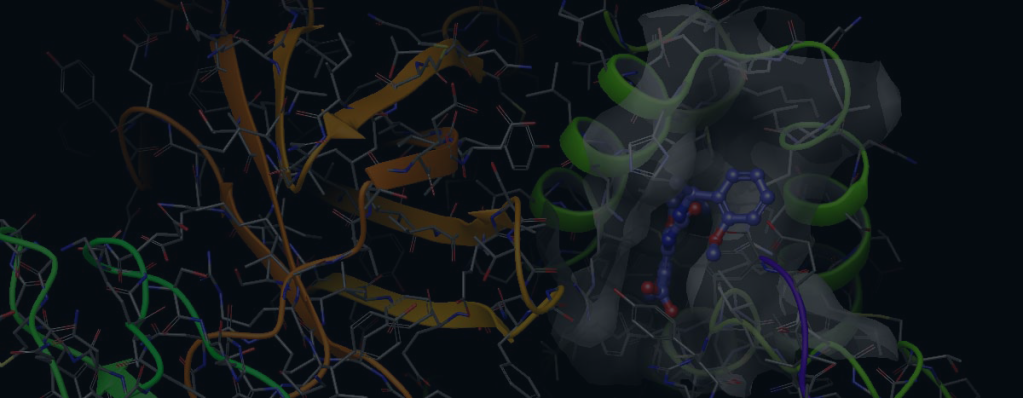
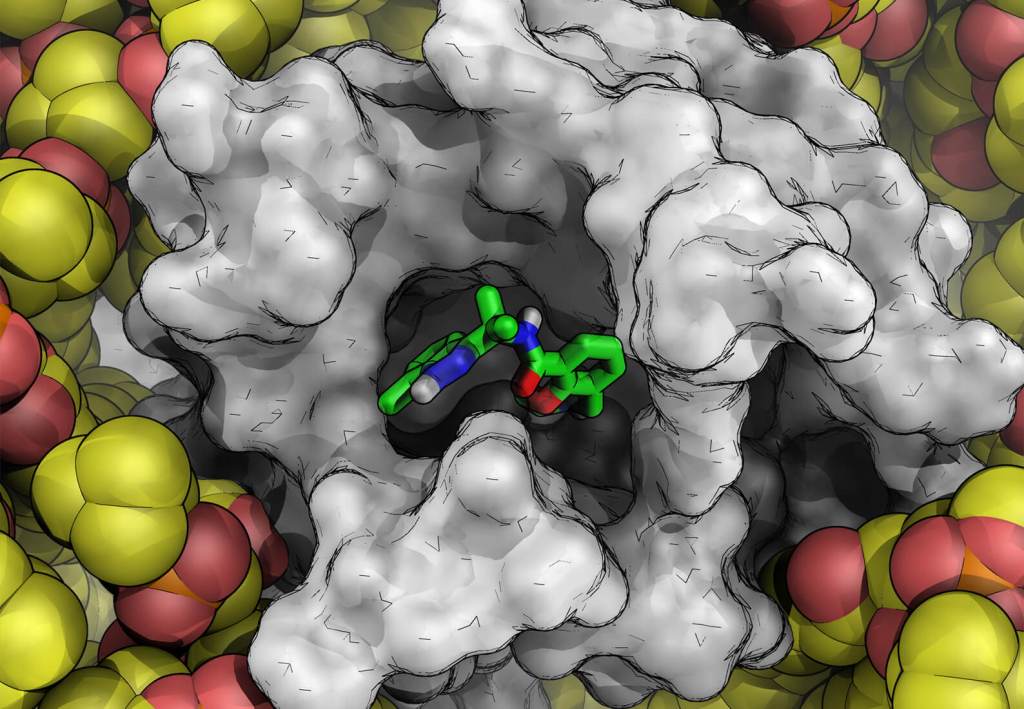
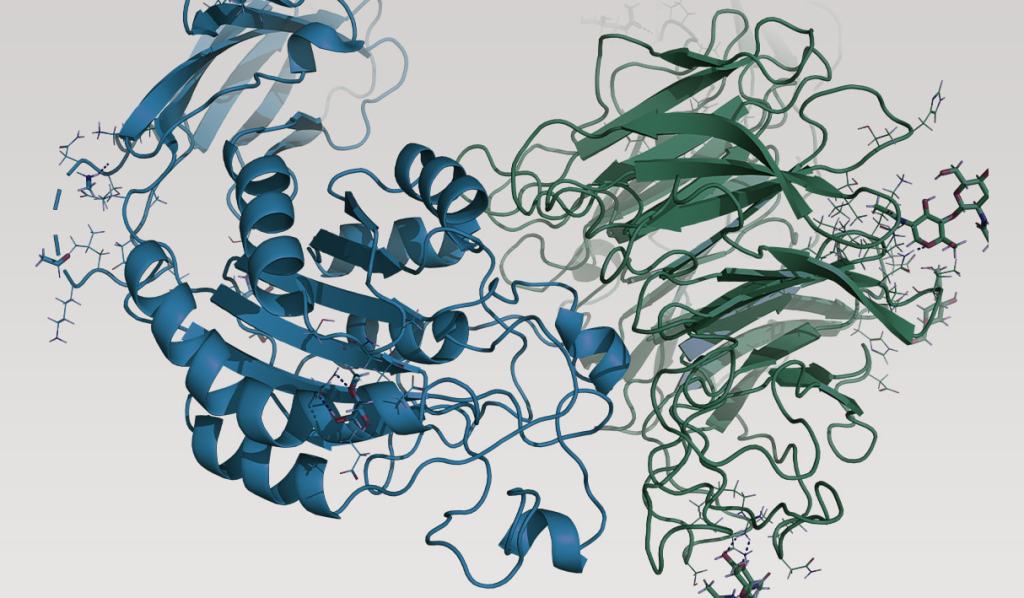
 Case Study
Case Study
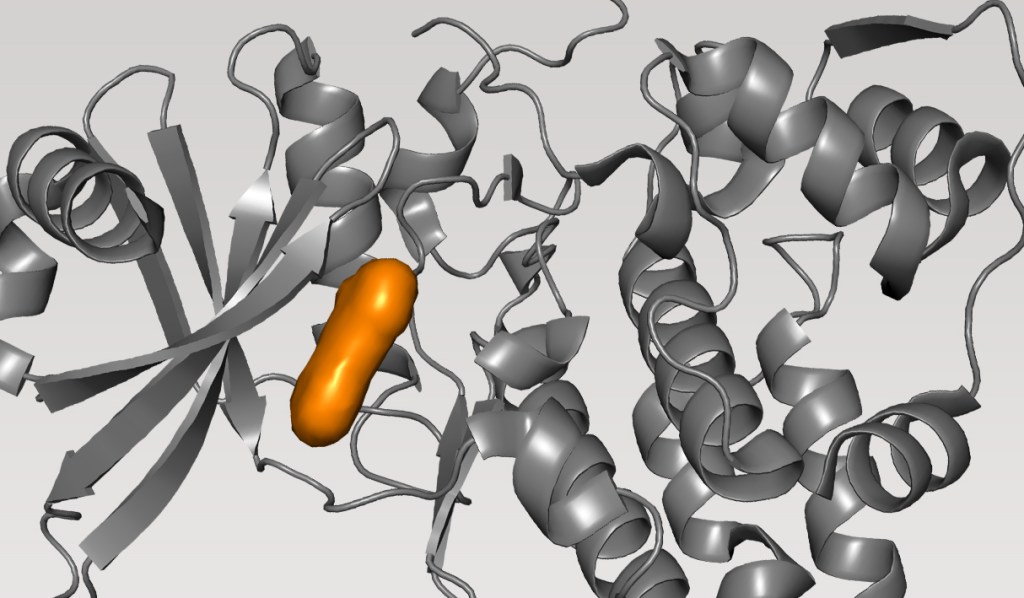 Case Study
Case Study
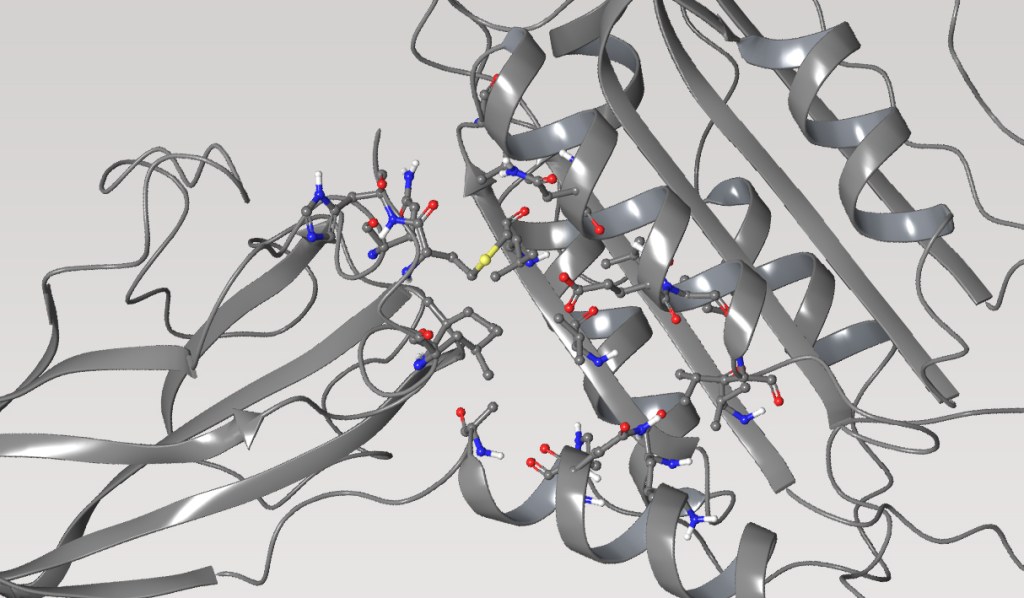 Case Study
Case Study
 Case Study
Case Study
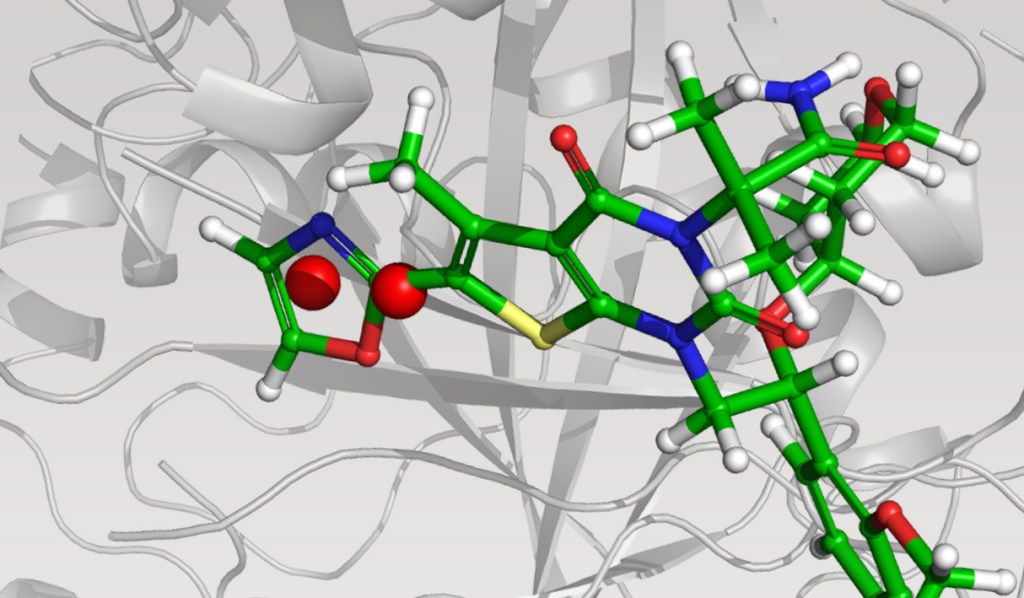 Case Study
Case Study
 Case Study
Case Study
 Case Study
Case Study
 Case Study
Case Study
 Case Study
Case Study
 Case Study
Case Study

Latest insights from Extrapolations blog
Training & Resources
Online certification courses
Level up your skill set with hands-on, online molecular modeling courses. These self-paced courses cover a range of scientific topics and include access to Schrödinger software and support.
Free learning resources
Learn how to deploy the technology and best practices of Schrödinger software for your project success. Find training resources, tutorials, quick start guides, videos, and more.
Other Resources