APR 29, 2026
Computational Visualization of Molecular Geometry, Bond Polarity, and Intermolecular Forces
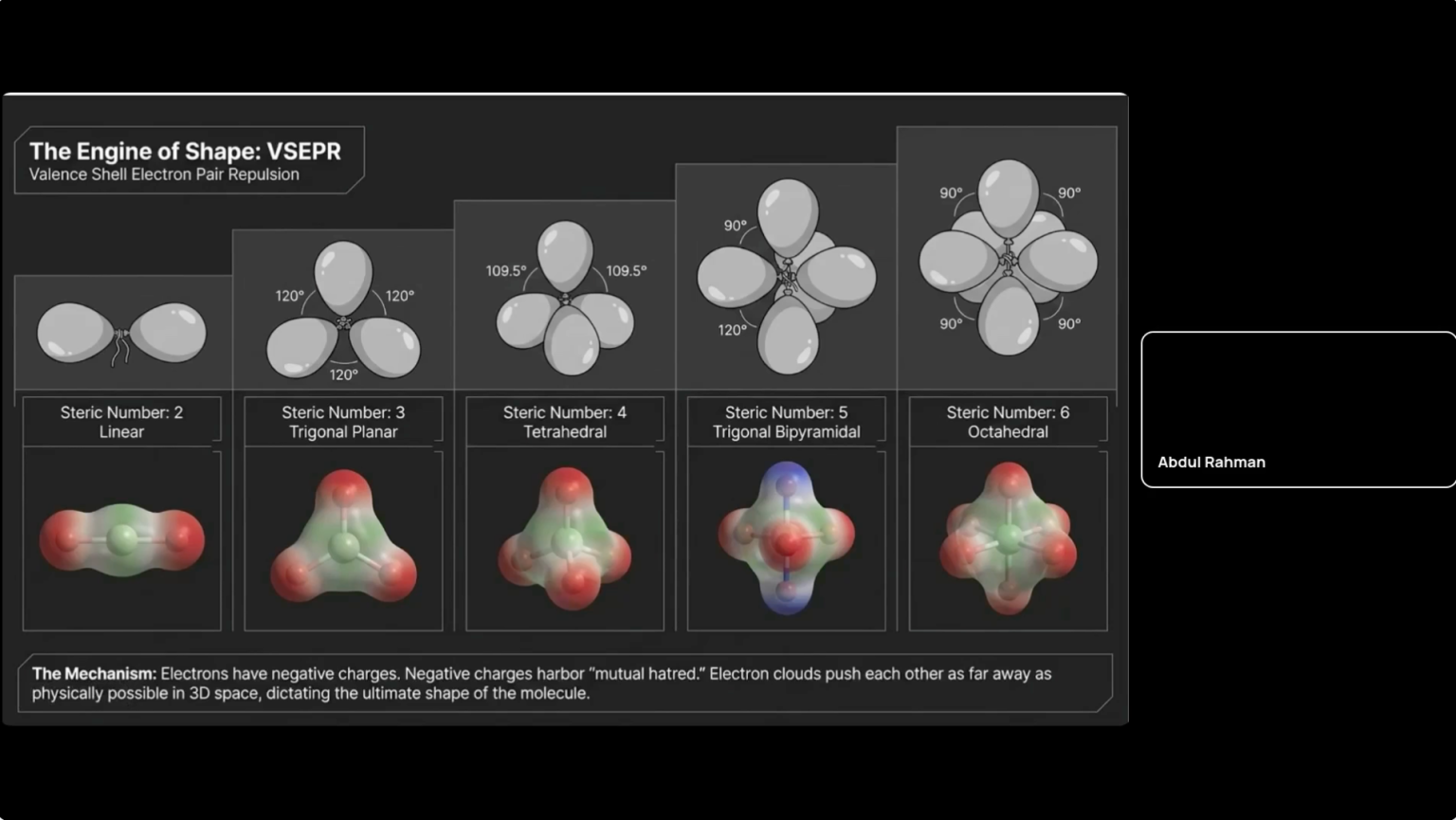
We present a computational study and teaching module that uses molecular modeling tools to illustrate fundamental structure‐bonding concepts in chemistry. Students build and visualize 3D structures (e.g. VSEPR-based shapes) using open-source graphics programs (Avogadro/IQmol) and quantum calculations. In a sample exercise, learners select template molecules covering VSEPR steric numbers 2–6 and modify atoms to generate new geometries. This approach automatically enforces correct bond angles for linear, trigonal, tetrahedral, etc., as in physical model kits. Students then compute and display electrostatic potential maps and partial atomic charges for each molecule. For example, Avogadro’s tools can color-code surfaces by electrostatic potential and draw a dipole-arrow on the structure. As Avogadro documentation notes, such electrostatic maps “help to visualize charge distribution” and thus directly reveal bond polarity. These visualizations make electronegativity differences and molecular dipoles explicit: bonds with large electronegativity gaps show large surface polarization and dipole arrows, whereas nonpolar molecules show symmetric potential surfaces.
We also extend the models to intermolecular forces: by simulating small clusters or dimers (e.g. water or ammonia pairs) using molecular mechanics or dynamics (Schrödinger Desmond, GROMACS, etc.), we animate hydrogen-bond formation and van der Waals attractions. In these simulations, electrostatic potentials on each molecule help explain dipole–dipole interactions, and trajectories illustrate transient contact (dispersion) forces. For instance, a water dimer model shows partial-charge regions aligning to form an H-bond, while gas-phase trajectories of nonpolar molecules highlight fleeting London forces. In practice, combining geometry visualization with electrostatic surfaces mirrors prior work in computational chemistry education: MolView-based modules and Avogadro projects have been shown to significantly improve conceptual understanding of bonding and polarity. We adapt these ideas for a chemistry conference presentation, showing screenshots of modeling exercises and discussing student outcomes.
Our Speaker

Abdul Rahman
Research Engineer
“Abdul Rahman is a Research Engineer and educator with over eight years of experience bridging the gap between traditional metallurgical science and modern artificial intelligence. Holding a Master’s degree in Metallurgical and Materials Engineering from UET Lahore, Abdul has cultivated a “”physics-first”” approach to Materials Informatics, ensuring that digital models are rigorously grounded in physical material behavior. Throughout his career, Abdul has combined experimental metallurgy with computational innovation—from optimizing iron ore beneficiation and developing biodegradable Mg-based implants to managing ISO 17025 laboratory systems. Recently, he has shifted his focus toward developing AI-driven tools, such as CorroScope-AI and agentic workflows, designed to manage complex datasets for predictive property modeling.”