Oct 8, 2025
Structure-based discovery of highly potent dihydroorotate dehydrogenase inhibitors for once-monthly malaria chemoprevention
We are delighted to bring you another webinar in our series Stories of Drug Hunting in a Digital Age – featuring conversations with veteran drug hunters who share their unique drug discovery stories, from idea to development candidate. You’ll hear about the teams and tools used to progress programs and have an opportunity to ask questions.
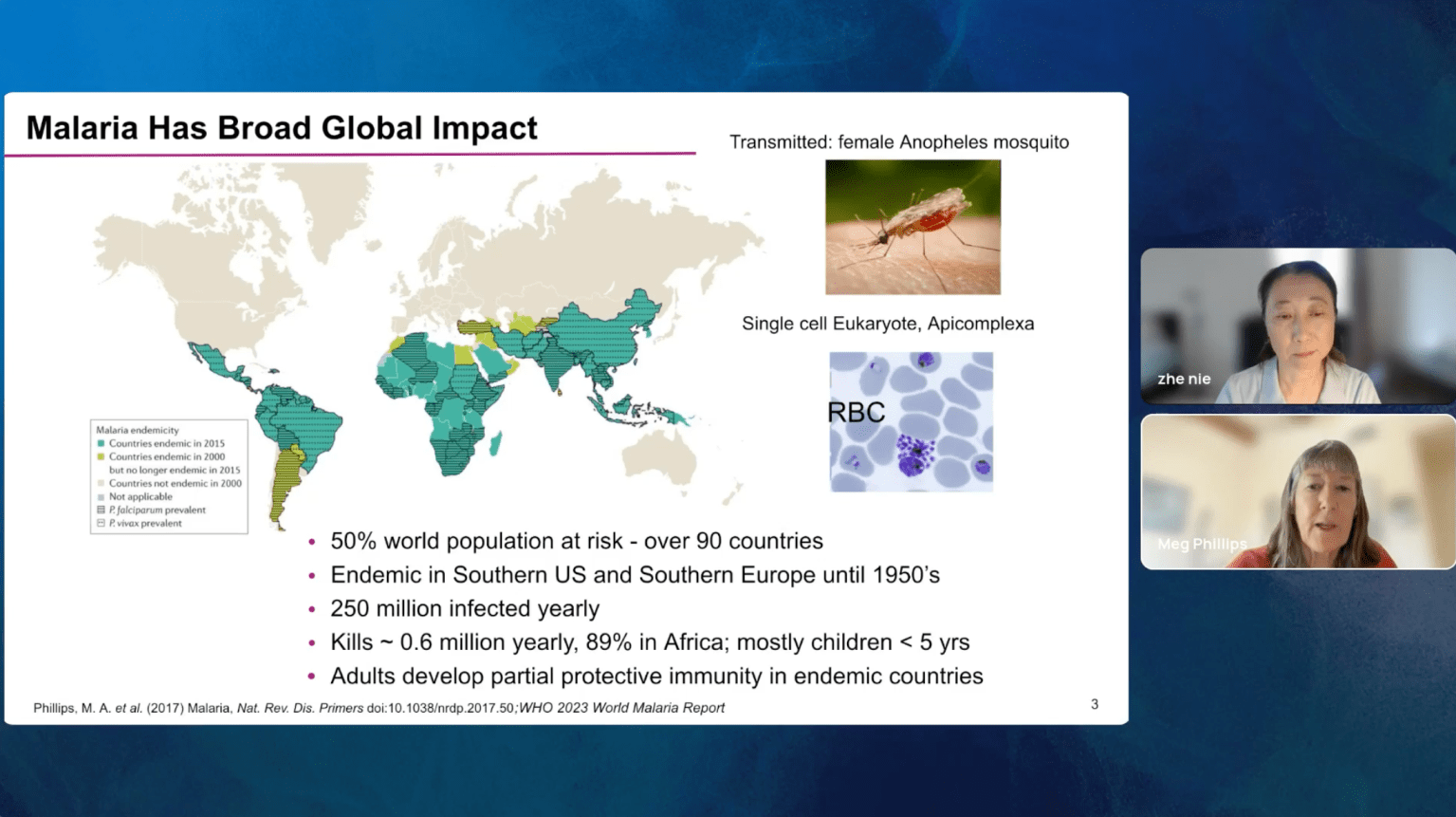
Malaria remains a serious global health challenge, yet treatment and control programs are threatened by drug resistance. Dihydroorotate dehydrogenase (DHODH) was clinically validated as a target for treatment and prevention of malaria through human studies with DSM265 (Phase 2), but currently no drugs against this target are in clinical use.
In this webinar, Zhe Nie, medicinal chemist and project leader at Schrödinger, and Margaret Phillips, professor at UT Southwestern Medical School, Dallas, share in conversation how their teams worked collaboratively towards the discovery of novel DHODH inhibitors. They will explore how structure-based computational tools including free energy perturbation (FEP+) were used to discover highly ligand efficient, potent, and selective pyrazole-based Plasmodium DHODH inhibitors through a scaffold hop from a pyrrole-based series. Optimized pyrazole-based compounds were identified with low nM-to-pM Plasmodium falciparum cell potency and oral activity in a humanized SCID mouse malaria infection model. The lead compound DSM1465 is more potent and has improved ADME/PK properties compared to DSM265. This compound meets Medicines for Malaria Venture’s objective of identifying compounds with potential to be used for once-monthly chemoprevention in Africa to support malaria elimination efforts.
Webinar Highlights
- Discover how in silico techniques combined with traditional medicinal chemistry approaches are applied in a structure-based drug discovery program
- See how the team overcome DMPK challenges to discover a pre-clinical candidate, DSM1465, with a long predicted human half life at a low dose
- Learn how free energy perturbation methods (FEP+) were used to prioritize compounds and drive programs forward efficiently
- Ask questions to gain further insight from the speakers to apply to your work
Publication
- Nie Z, et al. J. Med. Chem. 2025, 68, 1, 590–637.
Our Speakers

Zhe Nie
Executive Director, Medicinal Chemistry, Therapeutics Group, Schrödinger
Dr. Zhe Nie is the Executive Director of Medicinal Chemistry, Therapeutics Group, at Schrödinger. She has been leading multiple wholly owned and partnered drug discovery programs. Most recently, she led Schrödinger’s MALT1 discovery project team, successfully developed the small molecule drug SGR-1505 (Schrödinger’s first internal clinical asset currently in Ph1) by applying Schrödinger’s computational platform. It took less than two years from the start of the project to the selection of the clinical candidate. In collaboration with Dr. Philips from Southwestern Medical Center and other academic collaborators, Dr. Nie also played a key role in discovering a highly potent DHODH inhibitor as a potential once-monthly chemopreventive antimalaria treatment. She has extensive experiences applying advanced computational tools to assist in the design of small molecule drug candidates. She previously worked at Takeda, Celgene, and Quanticel Pharmaceuticals (acquired by Celgene), where she led and contributed to the advancement of multiple small molecule drugs to the clinics including TAK-960, TAK-659 and CC-90011.

Margaret A. Phillips
Professor and Chair, Dept of Biochemistry, UT Southwestern Medical School, Dallas
Meg Phillips is a parasitologist recognized for her work on exploiting metabolic pathways in protozoan parasites for drug discovery. Her work on the pyrimidine biosynthetic pathway in the malaria parasite led to the identification of an inhibitor of dihydroorotate dehydrogenase that reached clinical development for the treatment of malaria, for which she received the Medicines for Malaria Venture (MMV) Project of the Year Award for this work in 2010. She has identified additional inhibitors against this target with the potential to advance for chemoprevention and has also advanced efforts to target other key enzymes in the malaria parasite, including the proteasome. She has made multiple discoveries focused on the polyamine metabolic pathway in Trypanosoma brucei, the causative agent of sleeping sickness. She identified novel regulatory mechanisms for two enzymes in the pathway, S-adenosylmethionine decarboxylase and deoxyhypusine synthase, finding that both enzymes require oligomerization with inactive paralogs for activity. Phillips graduated from the University of California, Davis with a B.S. in Biochemistry in 1981, and with a Ph.D. in Pharmaceutical Chemistry in 1988 from the University of California, San Francisco, where she was also a postdoctoral fellow in the Department of Biochemistry. She joined the faculty of UT Southwestern in the Department of Pharmacology as an assistant professor in 1992, was promoted to tenured associate professor in 1997, and to full professor in 2002. She became Chair of the Department of Biochemistry at UT Southwestern in 2016 and holds the Sam G. Winstead and F. Andrew Bell Distinguished Chair in Biochemistry. She was elected to the National Academy of Sciences in 2021, elected as a fellow of the American Society for Biochemistry and Molecular Biology and elected to the fellowship of the American Academy of Microbiology in 2022. She won the 2024 ASBMB Herbert Tabor Research Award and the 2026 ASBMB Alice and CC Wang Award in Molecular Parasitology.