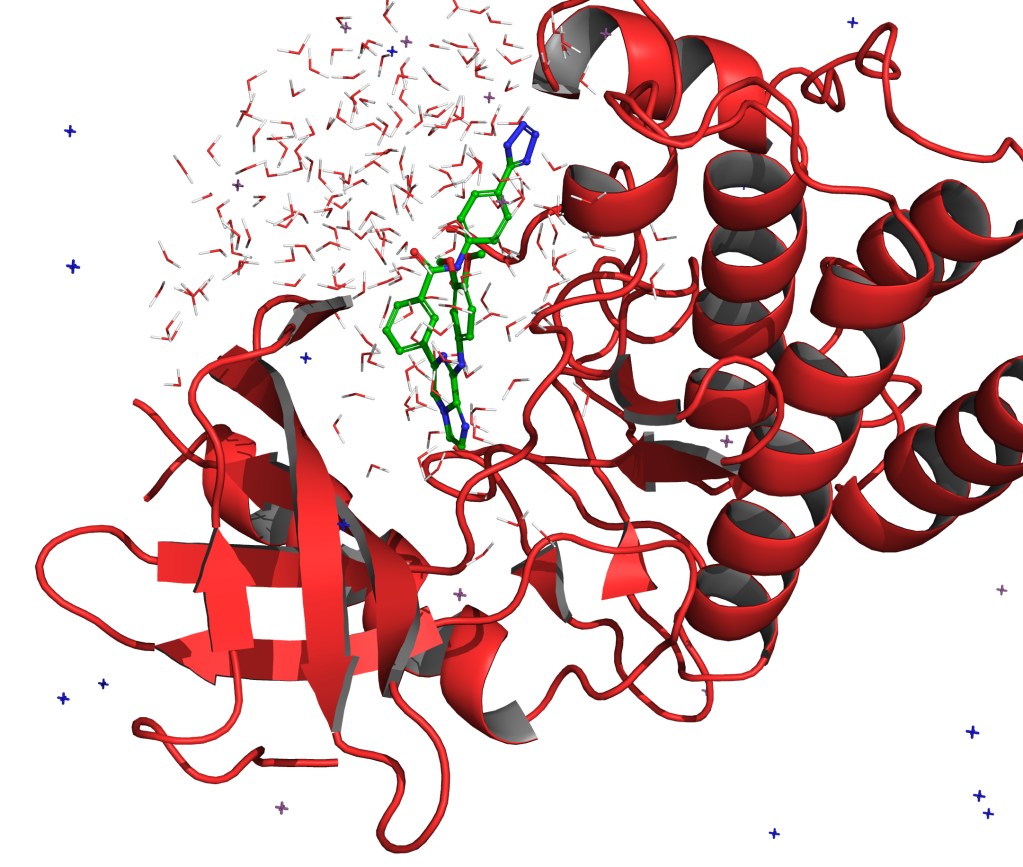
On Ternary Complex Stability in Protein Degradation: In Silico Molecular Glue Binding Affinity Calculations
Linking ATP and allosteric sites to achieve superadditive binding with bivalent EGFR kinase inhibitors
Structure-Guided Design of a Domain-Selective Bromodomain and Extra Terminal N-Terminal Bromodomain Chemical Probe
Discovery of an Oral, Beyond-Rule-of-Five Mcl-1 Protein–Protein Interaction Modulator with the Potential of Treating Hematological Malignancies
Discovery and Optimization of the First ATP Competitive Type-III c-MET Inhibitor
Structural and mechanistic insights into the inhibition of respiratory syncytial virus polymerase by a non-nucleoside inhibitor
Next-generation protein engineering with physics: Predicting affinity, stability, selectivity, and pH-dependent binding
FEB 5, 2025
Next-generation protein engineering with physics: Predicting affinity, stability, selectivity, and pH-dependent binding
The landscape of protein engineering is evolving rapidly, driven by cutting-edge computational techniques that are transforming biopharmaceutical discovery and optimization. Together, we will explore groundbreaking methods that enable accurate modeling of critical physical endpoints, including thermal stability, protein-protein binding affinity, and pH sensing profiles. These innovations promise to accelerate early-stage discovery while enhancing the precision of biotherapeutic design.
In this webinar, we will introduce Schrödinger’s FEP+ Residue Scan, a next-generation tool that achieves approximately 15x speedup compared to traditional Protein FEP methods, without significant loss of accuracy. This capability enables researchers to perform large-scale residue perturbation studies with high efficiency and reliability.
We will also present a practical example of computational antibody optimization, showcasing how these tools can be applied to real-world challenges. Additionally, we will discuss how Schrödinger’s biologics services can further streamline and customize workflows, empowering scientists to tackle complex projects effectively. Join us to discover how these next-generation solutions are transforming the field of protein engineering and driving impactful innovation in the biopharmaceutical industry.
Webinar Highlights
- Overview of Schrödinger’s biologics capabilities and offerings for rational antibody design
- Key features of FEP+ Residue Scan and Protein FEP+ for protein mutational engineering
- Introduction to Schrödinger’s biologics services to lower the barrier to entry for modeling novices
Our Speakers

Dan Cannon
Principal Scientist II, Schrödinger
Dr. Dan Cannon is a Principal Scientist within Schrödinger’s Applications Science team and the EU lead for biologics services in Europe. Prior to joining Schrödinger, Dan received his Ph.D. from the University of Strathclyde in Glasgow, UK, under the supervision of Prof. Tell Tuttle. In 2016, he began working at MedImmune (now AstraZeneca) in Cambridge, UK, using computational approaches for therapeutic protein design. Since joining Schrödinger in 2018, Dan has leveraged his extensive biologics expertise to enable Schrödinger customers to create and deploy cutting-edge computational workflows and design better molecules, faster.

Lingle Wang
Senior Vice President, Scientific Development, Schrödinger
Dr. Lingle Wang joined Schrödinger in 2012. He is responsible for advancing Schrödinger’s physics-based computational drug discovery platform. He obtained his Ph.D. from Columbia University working with Professors Richard Friesner and Bruce Berne on methods to quantify the role of water molecules in protein-ligand binding, enhanced sampling in biomolecular simulations, and free energy calculations. Lingle has published extensively in the areas of free energy methods development and applications in drug discovery.
Virtual testing of personal care and cosmetics formulations using digital chemistry methods
FEB 19, 2025
Virtual testing of personal care and cosmetics formulations using digital chemistry methods
持続可能な製品の開発には多くの課題があり、時間やリソース、新しい原材料が必要です。このプロセスを効率化するために、予測モデリングが注目されています。これにより、基準を満たす有望な成分や配合、新たな包装材料を特定でき、計算手法を用いた仮想試験を通じて分子レベルでの理解が得られます。具体的には、個々の成分の挙動や配合の形態、安定性、生体表面との相互作用を解析可能です。また、製品と包装材料の相互作用についても探究でき、保存期間に大きく影響する要因を把握できます。
本セミナーでは、ケーススタディを通じて、計算化学が製品開発、容器設計、製品使用時の解析にどのように役立つかを示します。
Our Speaker

Jeffrey M. Sanders, Ph.D.
Product Manager and Scientific Lead of Consumer Goods, Schrödinger
ウースター工科大学で応用物理学の学士号を取得後、トーマス・ジェファーソン医科大学で生物物理学および分子薬理学の博士号を取得しました。2013年にシュレーディンガー社に入社して以来、計算化学ソフトウェアの科学的および技術的な側面で複数の役割を担ってきました。現在、消費財分野のテクニカルリードおよび製品マネージャーを務めています。
Innovations in Protein Modeling: How Physics is Impacting Industrial Drug Discovery
NOV 7, 2024
Innovations in Protein Modeling: How Physics is Impacting Industrial Drug Discovery
In this webinar, hear from The Protein Society and invited speakers from Schrödinger, UCB, and Nested Therapeutics as they discuss three real-life drug discovery case studies that were impacted by the use of physics-based molecular simulations.
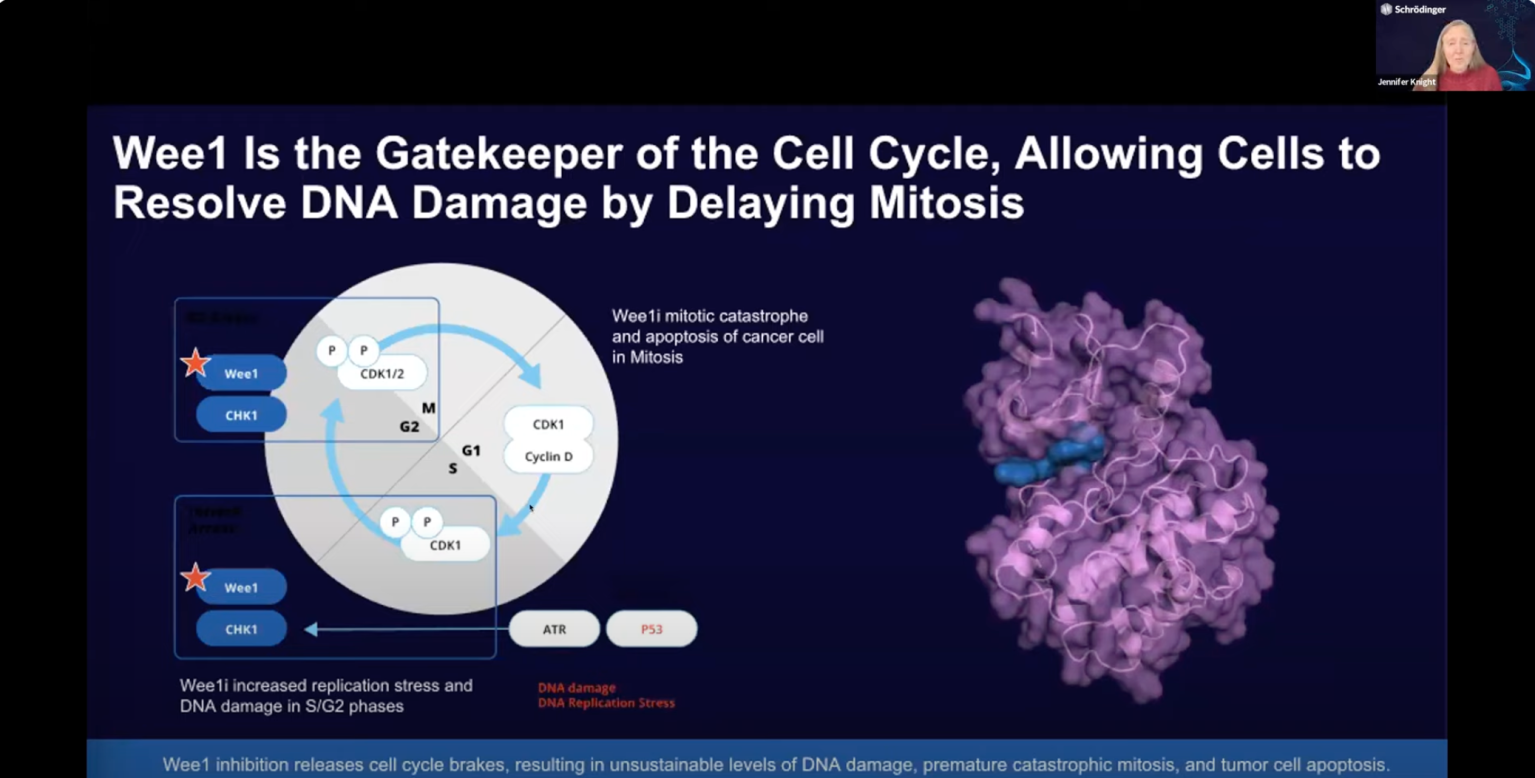
Achieving kinome-wide selectivity in drug discovery campaigns through free energy calculations: a Wee1 case study
Dr. Jennifer Knight, Schrödinger
Driving drug discovery by physics-based simulations: Multifaceted impact beyond chemistry prioritization
Dr. Aysegul Ozen, Nested Therapeutics
アーカイブ配信: OPLS5及び解離速度定数の予測技術のご紹介
アーカイブ配信: OPLS5及び解離速度定数の予測技術のご紹介
- December 18th, 2024
- Virtual
2024年には数多くの分子動力学シミュレーション関連の新機能がリリースされました。
本ウェビナーではそれらの中からOPLS5とunbinding kinetics workflowに焦点を当ててご紹介します。
OPLS5では分極電荷、及び電荷移動を考慮する機能が追加されました。電荷変化を取り扱う手法や精度検証の結果をご紹介します。
unbinding kinetics workflowはPath CVを用いたInfrequent Metadynamicsを用いてリガンド-レセプターの解離速度定数(koff)の予測をするための技術です。Path CVの生成にはRandom Acceleration MDが使用されます。背景理論や精度検証の結果をご紹介します。
このウェビナーの録画視聴をご希望の方は、こちらの申込書にご記入ください。
※ご質問、ご不明な点がございましたら下記までお問い合わせください。
シュレーディンガー株式会社 機能紹介ウェビナー事務局
E-mail: info-japan@schrodinger.com