ConfGen
Accurate and efficient conformational search solution

Accurate and efficient conformational search solution
ConfGen is a knowledge-based method that combines empirically derived heuristics and physics-based force field calculations to efficiently produce high-quality, diverse, low-energy 3D conformers of small molecules. It improves the recovery of bioactive conformations among generated conformers and drastically speeds conformer generation in ligand-based virtual screening.
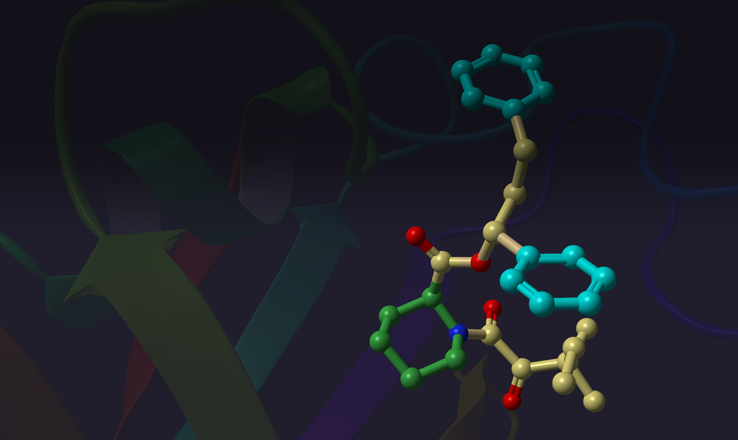
Given its exceptional speed and ability to reproduce low RMSD bioactive conformations, ConfGen is the ideal solution for generating conformations for ligand-based virtual screening. This white paper outlines the speed and accuracy of ConfGen for virtual screening applications.
Read the white paper
Learn more about the related computational technologies available to progress your research projects.
An easy-to-use pharmacophore modeling solution for ligand- and structure-based drug design
Efficient ligand-based virtual screening of millions to billions of molecules
Browse the list of peer-reviewed publications using Schrödinger technology in related application areas.
Level up your skill set with hands-on, online molecular modeling courses. These self-paced courses cover a range of scientific topics and include access to Schrödinger software and support.
Learn how to deploy the technology and best practices of Schrödinger software for your project success. Find training resources, tutorials, quick start guides, videos, and more.