Impacting drug discovery programs with large-scale de novo design
- January 25th, 2024
- 8:00 AM PT / 11:00 AM ET / 4:00 PM GMT / 5:00 PM CET
- Virtual
Developing technologies to more comprehensively and effectively enable de novo design of high-quality chemical matter has been a long-standing goal of drug discovery.
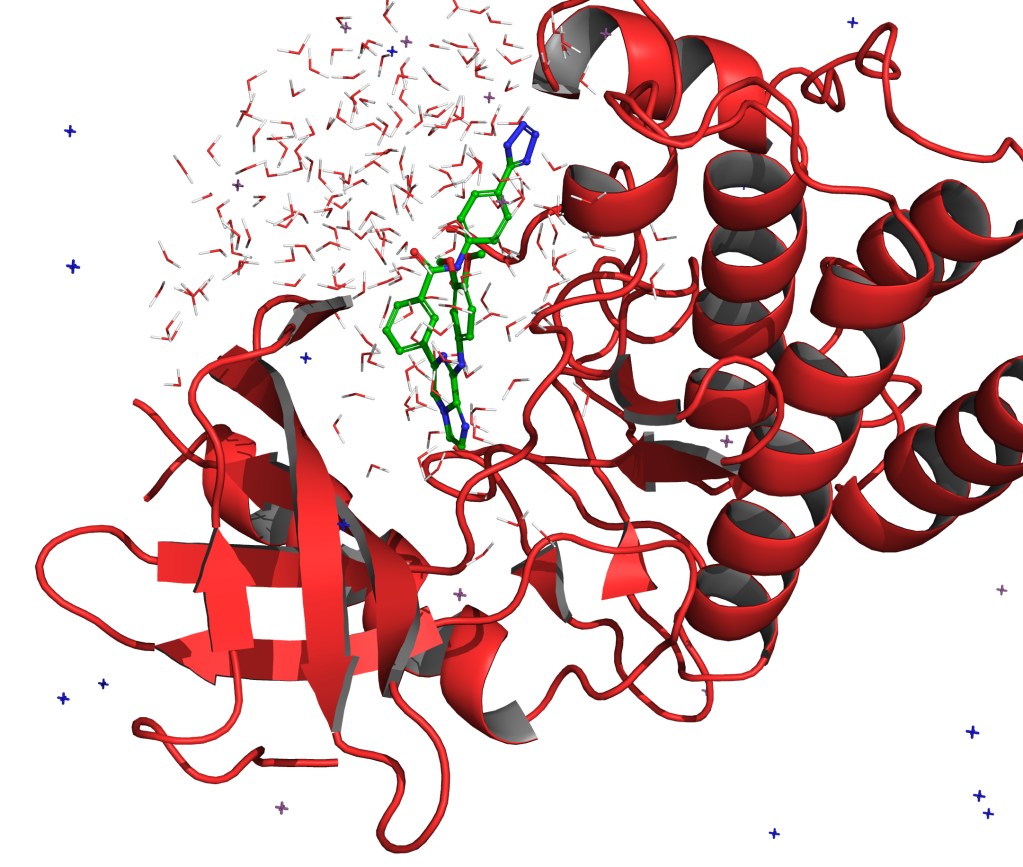
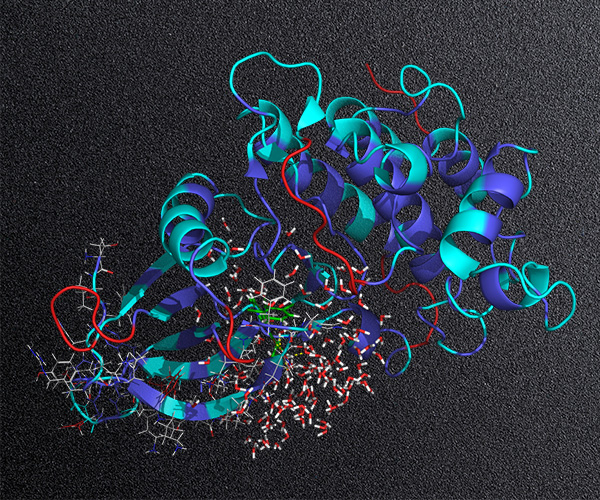
To this end, Schrödinger has recently spearheaded the development of workflows that combine large-scale synthetically-aware de novo design methods (AutoDesigner) with rigorous free energy-based scoring methods (Active Learning FEP+) for potency and selectivity optimization of small molecules. Recent developments of this technology move beyond R-group design to core exploration, enabling its expanded application to early stage hit identification efforts and the discovery of back-up series.
In this webinar, we will describe several recent case studies from Schrödinger’s therapeutics group where these de novo design technologies have allowed teams to overcome critical design challenges and accelerate programs.
Highlights
- Real-life comparison of AutoDesigner versus other common design methods, including an evaluation of chemical space explored, time spent, and ability to meet design goals
- Design of novel cores during hit identification using AutoDesigner
- Design of R-groups during hit-to-lead and lead optimization using AutoDesigner
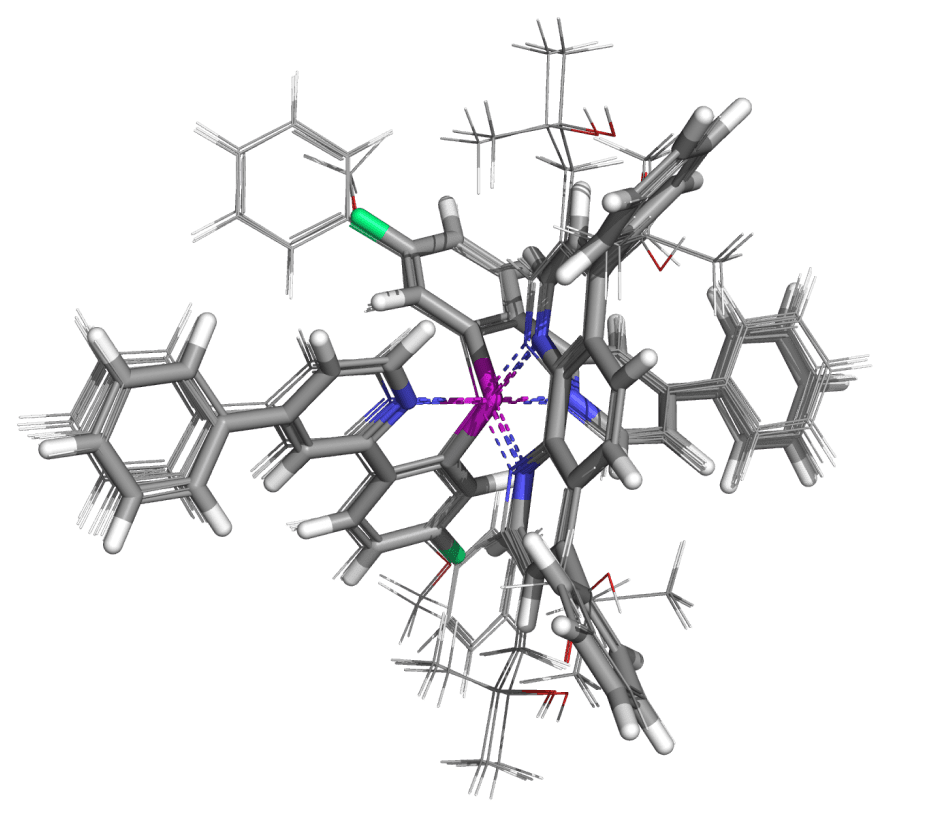
- Examples of improving potency and selectivity of a molecular glue and using de novo design to strengthen IP
- Requirements and best practices to apply the technology to your drug discovery programs
Impacting Drug Discovery Programs with Large-Scale De Novo Design
Our Speakers

Pieter Bos, PhD
Principal Scientist II, Schrödinger
Pieter Bos is a principal scientist and product manager of AutoDesigner and De Novo Design workflows. At Schrödinger, his main focus is the research, development and optimization of automated compound design algorithms. Lead scientist for the design and execution of enumerated drug molecule libraries for internal and collaborative drug design projects. He received his Ph.D. in Synthetic Organic Chemistry from the University of Groningen in the laboratory of Prof. Ben Feringa. Prior to joining Schrödinger, he worked as a postdoctoral researcher in synthetic methodology development at Boston University (Prof. John Porco and Prof. Corey Stephenson) and small molecule drug discovery at Columbia University (Prof. Brent Stockwell.).

Sathesh Bhat, PhD
Executive Director, Schrödinger
Sathesh Bhat, Ph.D., executive director in the therapeutics group, joined Schrödinger in 2011. He is responsible for overseeing computational chemistry efforts on internal and partnered drug discovery programs at Schrödinger. Previously, Sathesh worked at both Merck and Eli Lilly leading computational efforts in several drug discovery programs. He obtained his Ph.D. from McGill University, which involved developing structure-based methods to predict binding free energies. Sathesh has co-authored multiple patents and publications and continues to publish on a wide variety of topics in computational chemistry.

Zef Könst, PhD
Principal Scientist II, Schrödinger
Zef Könst is a principal scientist in the therapeutics group at Schrödinger where he is responsible for drug discovery project execution as a medicinal chemist. Zef joined Schrödinger in 2020 after working at Novartis and Nurix Therapeutics and has contributed to four compounds in clinical development. He received his Ph.D. from University of California, Irvine under Professor Vanderwal.